Tasks for study designers in Oracle Life Sciences Central Designer
- 1. Create the Adverse Event forms
- 2. Create other forms to send to Oracle Argus Safety
- 3. Create a Safety Case form
- 4. Add Argus Safety integration-specific rules
- 5. Map Oracle InForm data items to Oracle Argus entities
- 6. Generate the Annotated Study Book
Tasks for users in Oracle Clinical One Digital Gateway
- 7. Set up SFTP server and resources
- 8. Create an SFTP credential key
- 9. Create an integration group
- 10. Create the test integration
- 11. Test the integration
- 12. Create a production integration
- 13. Monitor an integration
Tasks for sponsor administrators in Oracle Life Sciences InForm Publisher and Oracle Argus Safety
Tasks for users in Oracle Central Designer, Oracle InForm, and Oracle Argus Safety
Tasks for study designers in Oracle Central Designer
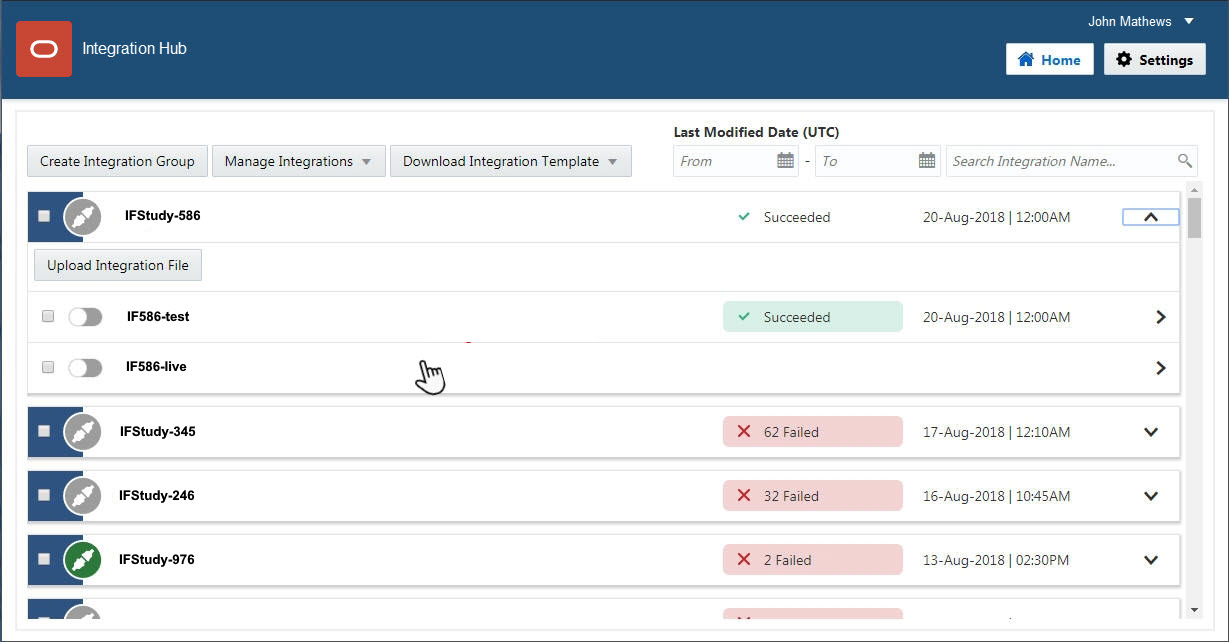
1Create one or more Adverse Event forms
When do I do this? At any time during study design, as long as the Safety and Data Management teams have agreed on what data should be sent from InForm to Argus.
Why should I do this? To collect adverse event data, which will be sent to Argus Safety.
You can design the study so that all the adverse event information is on a single Adverse Event form. Or, you can create multiple Adverse Event forms if you have different versions of the form; for example, for pregnancy or other types of adverse events.
Create an Adverse Event form.
Mark the form as Repeating. An Adverse Event form cannot be a common form.
Add adverse event items as needed, including the following required items:

A date/ time item to capture the onset date of the adverse event.

A Yes/ No item that triggers rules to initiate transmission of the adverse event data.
- Serious (the item will be reported). The question might be: Was this a Serious Adverse Event?
- Reportable (the item is not serious but should still be reported). The question might be: Should this event be reported?
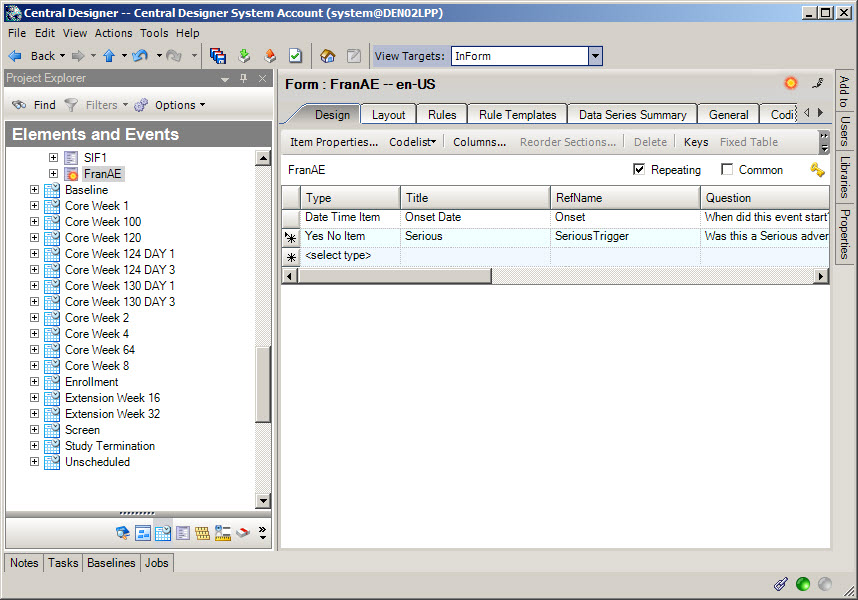
2Create other forms to send to Argus Safety
When do I do this? After you have created the Adverse Event form.
Why should I do this? This is how you send historical or clinical data to Argus as part of a case (for example, medical history, concomitant medications, lab test results).
Create one or more forms with data to send to Argus Safety.
For each form either mark the form as repeating, or leave the form as a flat form and create one or more repeating sections on it. Do not mark the form as Common.
In the form or section(s), add items that capture data related to the adverse event.

If you are using fixed repeating sections, you must assign codelists to the fixed items so that the site user can answer them.
When the site user submits these forms, Oracle InForm Publisher collects the adverse events and sends them to Oracle Argus Safety according to rules you define on the form, or using before and after onset dates configured in Oracle InForm Publisher.
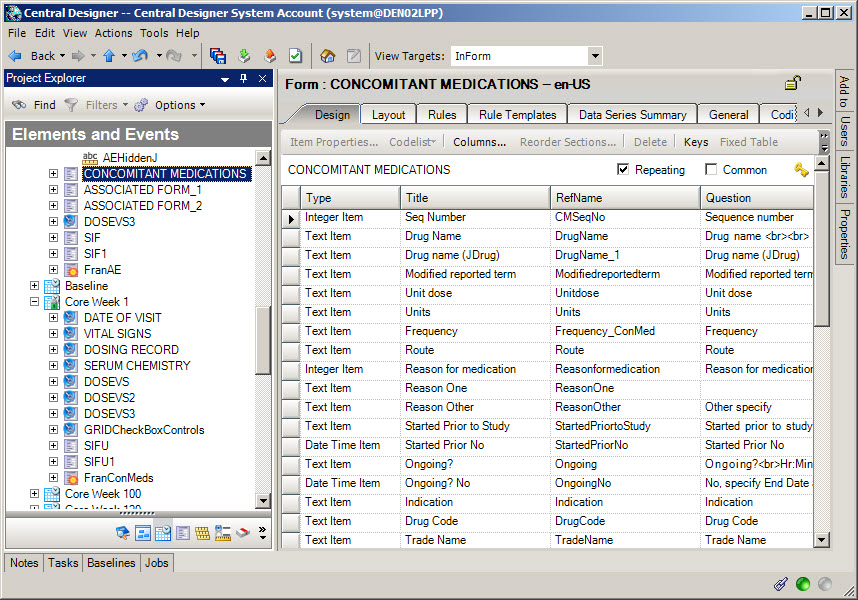
3Create a Safety Case form
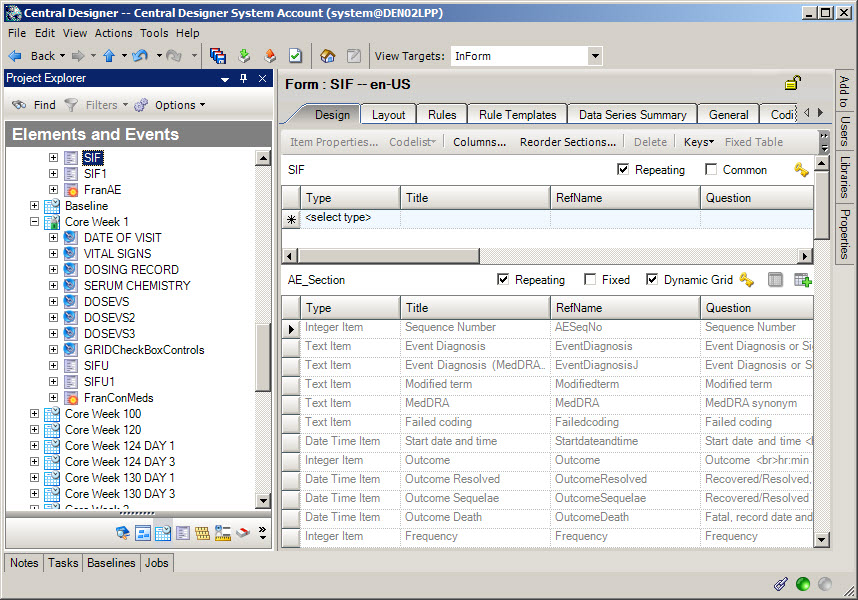
When do I do this? After you have created the Adverse Event form and other relevant forms to send to Argus Safety.
When do I need a Safety Case form? If you want Oracle InForm site users to be able to group multiple adverse events that relate to a single Argus case and report them together, you must include a Safety Case form. If each Adverse Event form becomes an Argus case, or you have configured Oracle InForm Publisher to send adverse events automatically based on their date of occurrence, you don't need a Safety Case form. The site user selects the items to send on the Safety Case form.
Create a Safety Case form.
Mark the form as Repeating.
Create a non-repeating section and add items that relate to the Argus case as a whole. You might include an item that triggers the case to be sent to Argus when the user submits the form by adding a question such as "Is this item ready to be sent to Argus?"
Create a repeating Adverse Event section in which the site user will identify the specific adverse events that they want to group together and submit as a single Argus case. When you create the Adverse Event section on the Safety Case form, create a dynamic grid that groups the adverse events by selecting the Dynamic Grid checkbox. Add the adverse event items to the Dynamic Grid by clicking the Dynamic Grid button at the top of the workspace and moving the items you want to include from the Available items for source list to the Selected items list.
Create repeating sections for other relevant information (Concomitant Medications, Medical History, Labs, etc.) that should be grouped together and submitted as a single Argus case. Create a dynamic grid that groups the other items.
4Add Argus Safety integration-specific rules
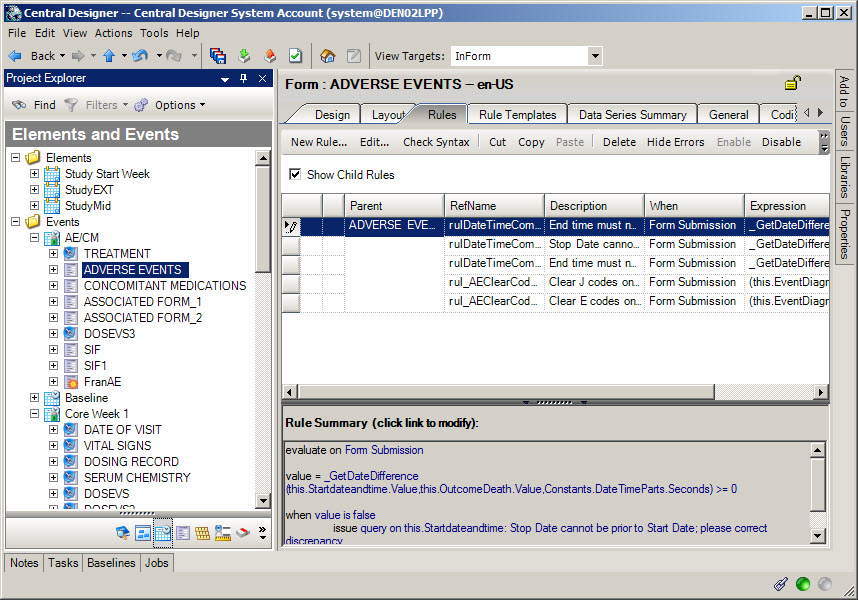
When do I do this? After the forms are complete.
Why should I do this? So that safety data can be sent from Oracle InForm to Oracle Argus.
Select the Adverse Event or Safety Case form, open the Rules tab, and create a new rule.
On the Preconditions tab, for Evaluate on Event, choose Form Submission.
On the Expression tab, enter the rule itself which specifies when to send the adverse event data to the Oracle InForm Publisher queue.
On the Actions tab, specify when to execute the rule. Either specify a value that must be True, or choose Always.
Rules for the Adverse Event form:

(Required) One of the following rules:
- Send the safety event, marked as Reportable or Serious on the Adverse Event form, to Argus Safety only when the Oracle InForm site user responds Yes to the "Ready to Send?" question.
- Send the safety event, marked as Reportable or Serious, immediately upon form submission without asking the Oracle InForm site user.
Note: If you have both an Adverse Event form and a Safety Case form, the rule you choose goes on the Safety Case form.

(Recommended if you are using a Safety Case form) Alert the Safety group or study team via email that there is a serious or reportable adverse event before the Oracle InForm site user completes the Safety Case form.
Rules for the Safety Case form:

If no adverse events are on the form, issue a query to alert the Oracle InForm site user to add an adverse event to the form or delete the entire form if the case is not needed anymore.
Rules for other forms containing adverse event items:

If there are two places where date of death or autopsy could be reported, check to make sure they are the same and, if not, create a query.
Note: For all other item-level rules that issue a query (for example, a check on onset date), you should reference the form and section as well as the item in the rule in the rule expression. For example, if you reference the full path on the onset date item on the Adverse Event form in a query rule, the query only fires on the Adverse Event form. If only the shared item is referenced, the query opens on both the Adverse Event form and Safety Case form, as the data is the same.
5Map Oracle InForm data items to Oracle Argus entities
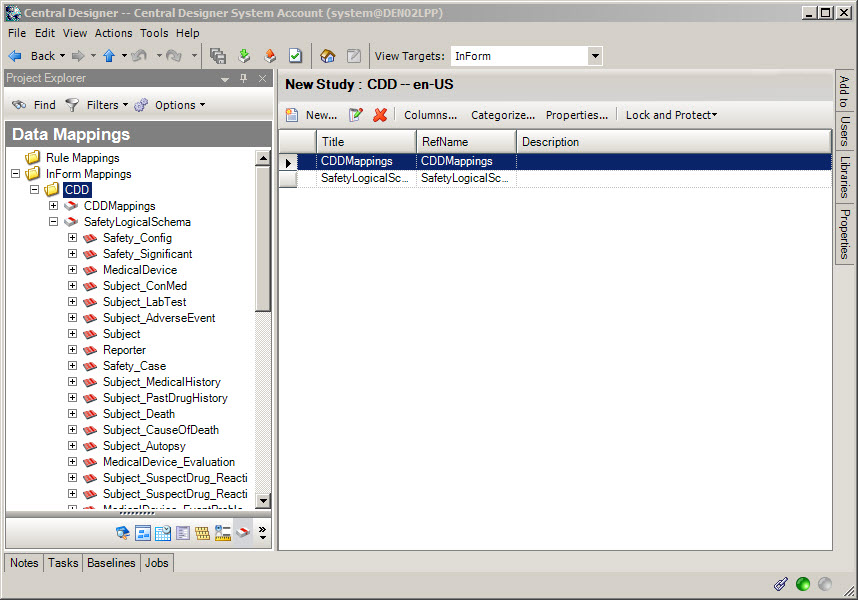
When do I do this? After you have created the forms.
Do I have to do this? Yes, Oracle InForm and Oracle Argus Safety use the data mapping feature of Oracle Central Designer to configure how safety event data items on Oracle InForm forms correspond to safety event entities in Argus Safety.
Download SafetyLogicalSchemaLibrary.csml from My Oracle Support (Doc ID 2301999.1). The file is an attachment in the bottom left corner of the Oracle Central Designer Release Notes page.
Import the file into your Oracle Central Designer study.
Start with the Adverse Event form.
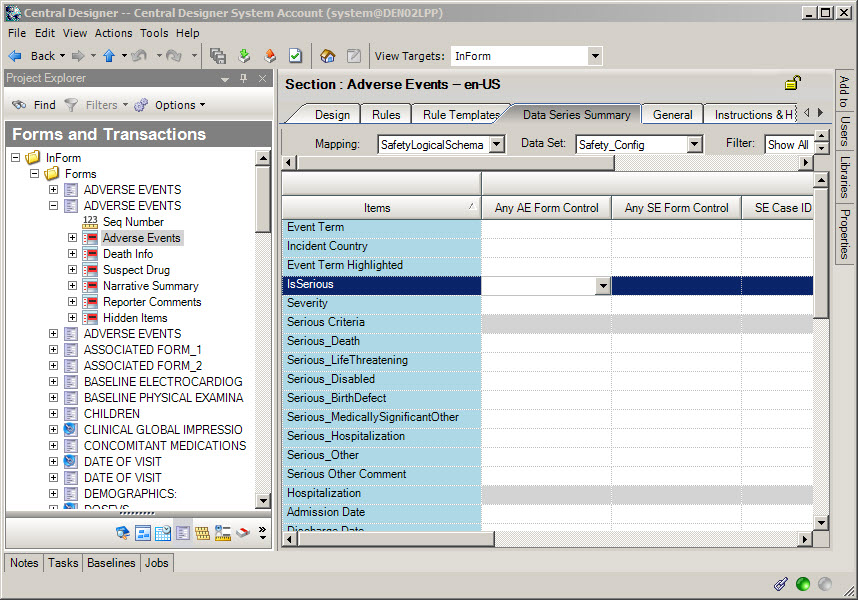
Add all items and forms to be sent to Argus to the Safety_Config data set. On the Data Series Summary tab, select SafetyLogicalSchema from the Mapping drop-down and Safety_Config from the Data Set drop-down. For each item, click the arrow at the right end of the data series cell and select Always to add this mapping on every form, study, and the library, or Form, to only map the item when it appears in this section on the form.
Change to the Safety_Significant data set and create mappings for those items you want Oracle InForm Publisher to monitor for changes and send a follow-up transmission to Argus when changes occur.
Perform the same two steps for the Safety Case form and any other forms with adverse event items on them.
All data mapped in Safety_Config will be sent to Argus when an update is triggered.
6Generate the Annotated Study Book and provide it to the integration manager
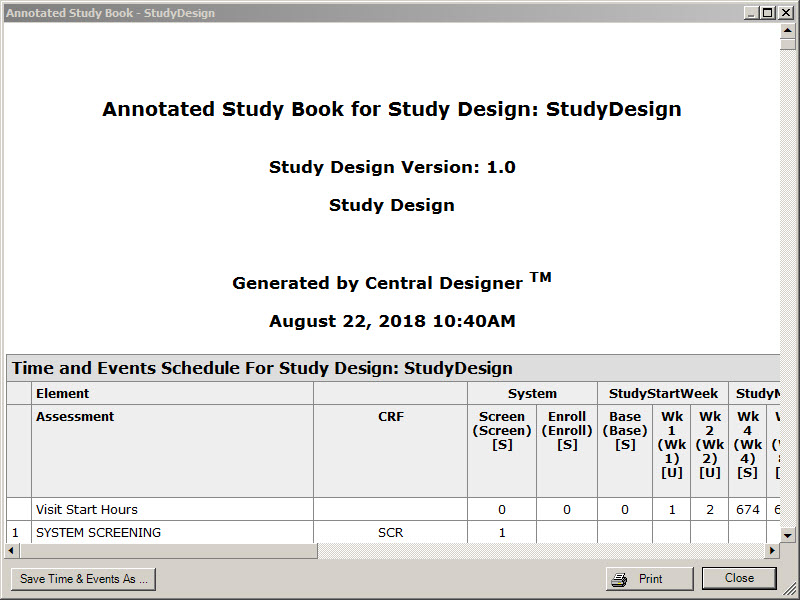
When do I do this? After the study design is complete.
Why should I do this? So that the integration manager knows what needs to be configured in Oracle Clinical One Digital Gateway.
Create an Annotated Study Book, and then print the book to a PDF file.
Provide the Annotated Study Book to the integration manager.
Tasks for users in Oracle Clinical One Digital Gateway
7Prepare the SFTP account and resources
Who does this? An integration manager.
When can I start working in Oracle Clinical One Digital Gateway? Once you do the following tasks, you are good to go:
Contact an Argus administrator and request an account for the Oracle HSGBU hosted customer SFTP server. You need this account to create a credential key.
Log in to the server with your SFTP account, create the necessary folders to store the E2B+R2 files for Argus Safety, and note the path to each folder from the server home. You can create one folder for the test integration, and one for the production integration, but you don't have to.
Contact an Argus administrator to get the Argus StudyID (which is case sensitive), the trade name of the products from the Argus product, and the license configuration in Argus.
Contact an Oracle Central Designer study designer and request the Annotated Study Book for the study you are integrating. You need it to get the study name and to map codelist values from Oracle InForm to the expected values in Oracle Argus Safety as part of setting up the integration.
8Create an SFTP credential key
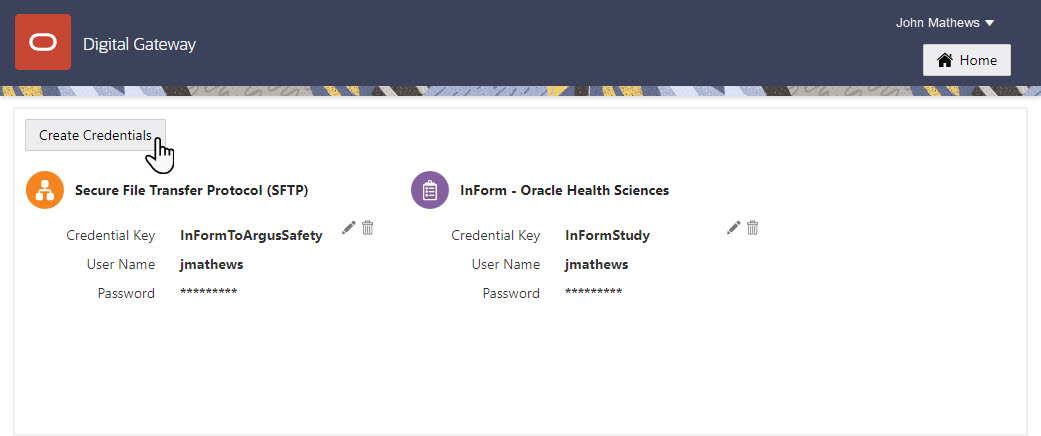
Who does this? An integration manager.
Why should I do this? SFTP credential keys are required to run a safe and proper integration between Oracle InForm and Oracle Argus Safety.
When do I do this? After you request the account on the SFTP server, set the folder where Oracle Clinical One Digital Gateway places the E2B+R2 files, get the Annotated Study Book, and have a list of all Oracle InForm fields that contain partial dates.
On the Home page, go to Settings and click Create Credentials to create your credential key for the SFTP server.
9Create an integration group for the study
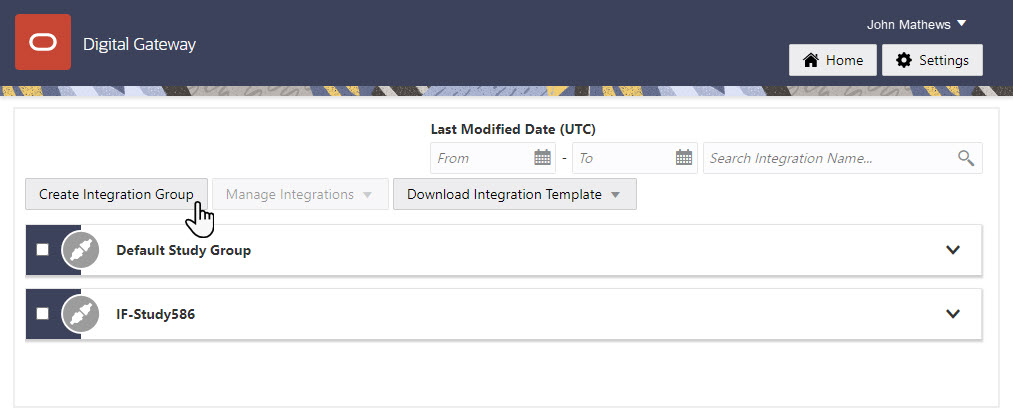
Who does this? An integration manager.
Why should I do this? For every Oracle InForm study that you integrate, you typically have at least two integrations: a test integration and a production integration. You can also have other integrations set up for the same study, such as a Oracle Clinical One Platform to Oracle InForm integration. Integration groups are a way to keep these integrations grouped together by study so that you can easily find and manage them.
When do I do this? After you create your SFTP credential key.
Click the Create Integration Group button to create an integration group for the Oracle InForm study.
Tip: We recommend that you include the Oracle InForm study name in the integration group name to help identify integrations for monitoring.
10Create and enable the test integration
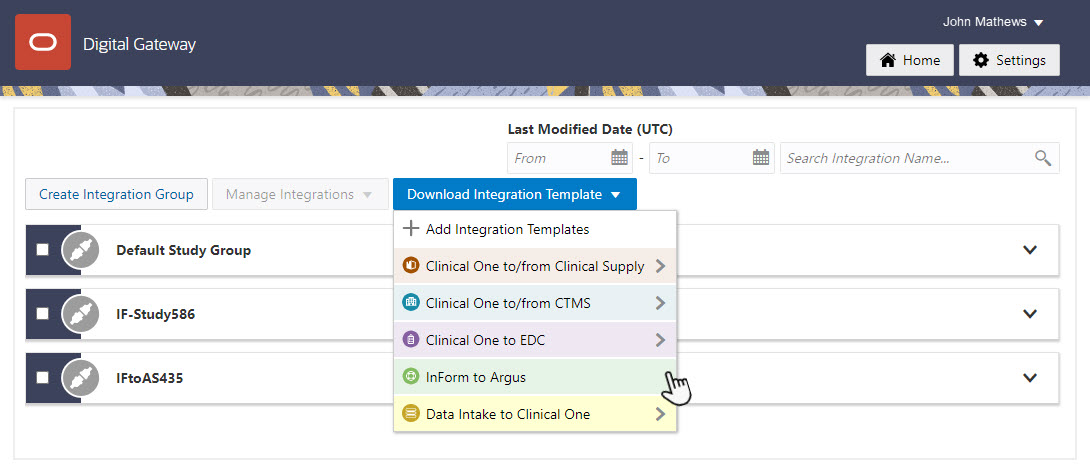
Who does this? An integration manager.
Why should I do this? You cannot test using a production integration, so you should create a separate integration for testing purposes.
When do I do this? After you create the credential key and an integration group in the UI.
On the Home page, download the template for the Oracle InForm to Oracle Argus integration.
Open the integration template in any editor, update it, and save it.
Find the integration group that you created, and upload the file.
Enable the test integration by clicking the toggle button next to it.
next to it.
Tip: If you want to add multiple integrations in one integration group, you need to upload a separate integration file for each integration.
Read step-by-step instructions on how to create the configuration file for a test integration.
11Test the integration
Who does this? An integration manager.
Why should I do this? To make sure the integration sends data as expected.
When do I do this? After you've uploaded the integration file and enabled the test integration file.
Work with SaaS Services to make sure that the test integration is running as expected.
When you are satisfied with the results, proceed to create the production integration based on the test integration.
12Create and enable the product integration
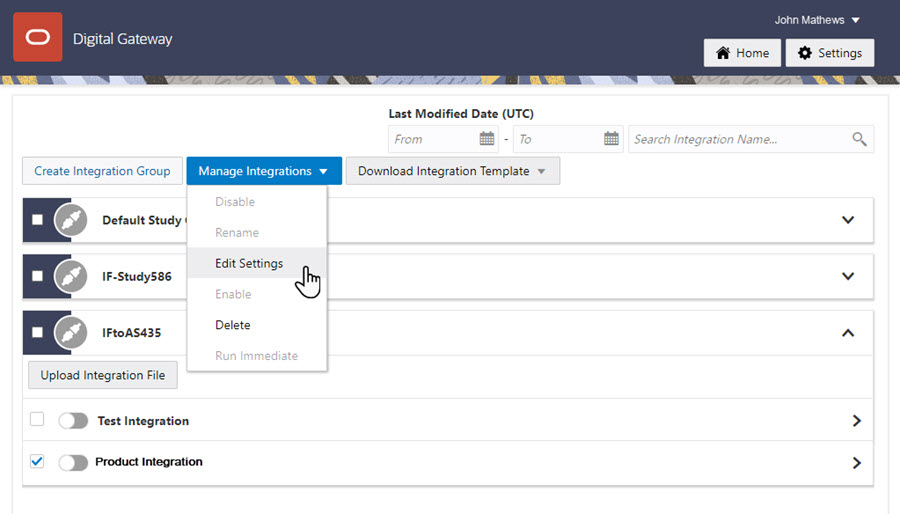
Who does this? An integration manager.
Why should I do this? So you can start sending safety case data from Oracle InForm to Oracle Argus Safety.
When do I do this? After you've monitored your testing integration and are satisfied with the results, and after the connection to the Argus Interchange has been set up.
Tip: Reuse the test integration file that you carefully updated to create the production integration.
On the Home page, click the down arrow for the integration group you created for the study.
Select the checkbox to the left of the test integration that you want to use as the base for this integration.
Along the top, click Manage Integrations, and click Edit Settings.
Download the test integration file, and save it locally under a representative name.
Open the file, and edit the SFTPRoot value and Argus study ID.
Go back to Oracle Clinical One Digital Gateway and look for the integration group that you created.
Expand it, and upload this file for your production integration.
Enable the integration.
Read step-by-step instructions on how to create a production integration.
13Monitor an integration
Who does this? An integration manager or an integration viewer.
Why should I do this? There are several use cases for integration failures and you should be aware of them. Unmapped data in the integration file can be one of the most common cases. Keep an eye on your integrations and jobs to make sure that everything is running smoothly.
When do I do this? After you upload your integration file in the right integration group and you enable the integration.
To check the status of a job or a safety report (after the integration is enabled), click the integration.
Read step-by-step instructions on how to monitor, edit, or troubleshoot an integration.
Tasks for sponsor administrators in Oracle InForm Publisher and Oracle Argus Safety
14Configure the Argus Safety-only parameters in Oracle InForm Publisher
When do I do this? After completing all previous tasks in this document.
Why should I do this? To set up how Oracle InForm Publisher moves the adverse event data from InForm to Argus Safety.
Install Oracle InForm Publisher on the Oracle InForm application server, as described in the Oracle InForm Publisher Installation Guide.
Identify the Argus Safety-related attributes that apply to the study.
Tips:

To configure time frames to trigger Oracle InForm Publisher to send safety data automatically, specify the number of days before and after the onset date of the adverse event (AE Range Starting Offset (days) and AE Range Ending Offset (days)) that Oracle InForm Publisher should use to include all Adverse Event forms started during the range or to include other forms associated with an adverse event.

If you are allowing the Oracle InForm site user to choose which AEs are relevant for sending to Argus Safety, instead of using time frames, you need to set AE Range Starting Offset and AE Range Ending Offset values to -1.

If the Oracle InForm site user can select relevant concomitant medications, set ConMed Range Starting Offset and Ending Offset to -1.

If the Oracle InForm site user can select relevant labs, set Lab Range Starting Offset and Ending Offset to -1.
When you submit a ticket to Oracle Services to set up the integration with Argus Safety, communicate which attributes to include and their values.
Oracle creates the subscriber and populate its settings, and then adds the trial to the subscriber.
15Set up the connection to Argus Interchange for Argus Safety
Who does this? A sponsor administrator.
When do I do this? After you receive your SFTP credentials and before you enable the integration in Oracle Clinical One Digital Gateway.
Why do I do this? So you can get safety cases from SFTP to Argus Interchange.

If your organization hosts Argus Safety, configure Axway or Oracle B2B so that files are pulled from the SFTP site into Argus Interchange.

If Oracle hosts Argus Safety, you don't need to do anything. Oracle B2B will be configured for you in your cloud environment so that files are pulled from the SFTP server into Argus Interchange.
Tasks for users in Oracle Central Designer, Oracle InForm, and Oracle Argus Safety
16In Oracle Central Designer, deploy the study to Oracle InForm
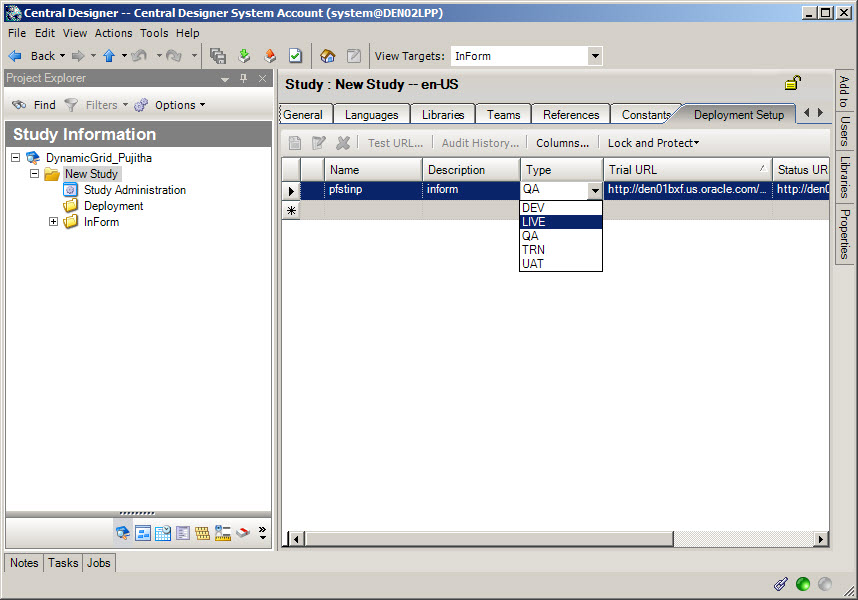
Who does this? An Oracle Central Designer study designer.
When do I do this? At any time after the study design is complete and the study has been provisioned.
Validate the study to create a deployment package, and then initiate deployment. During deployment, the study design moves directly from Oracle Central Designer to Oracle InForm.
17In Oracle InForm, complete the applicable safety forms
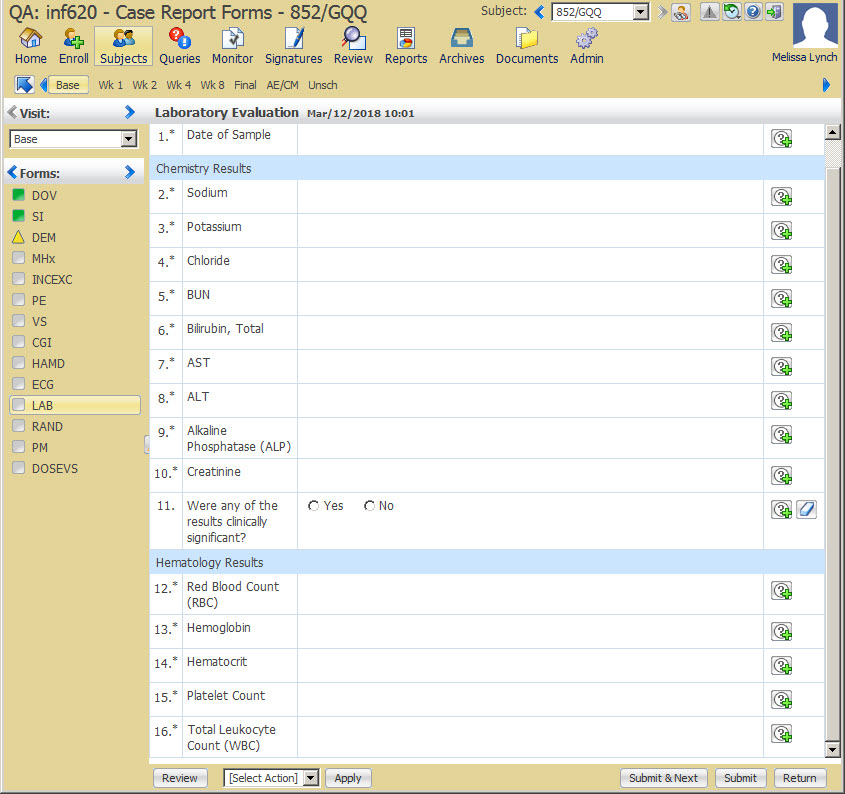
Who does this? An Oracle InForm site user.
When do I do this? As you meet with subjects.
Enter each adverse event for a subject on a separate Adverse Event form.
If the study was built with a Safety Case form:
Select the other instances of the Adverse Event form to send to Argus. For example, if the subject has had five adverse events and you think three should be part of the case, select the three adverse events in the dynamic grid by clicking the plus sign icon on the right.
Do the same for the other forms, such as Medical History, Concomitant Medications, Labs. For example, if there are 15 concomitant medications entered, but only 1, 4, 7, and 13 apply to the case, select the four medications in the dynamic grid by clicking the plus sign icon on the right.
After you have entered the required information and are ready to send the case to Argus, respond Yes to the Send to Safety question if it is present.
18In Argus Safety, verify that each case comes in
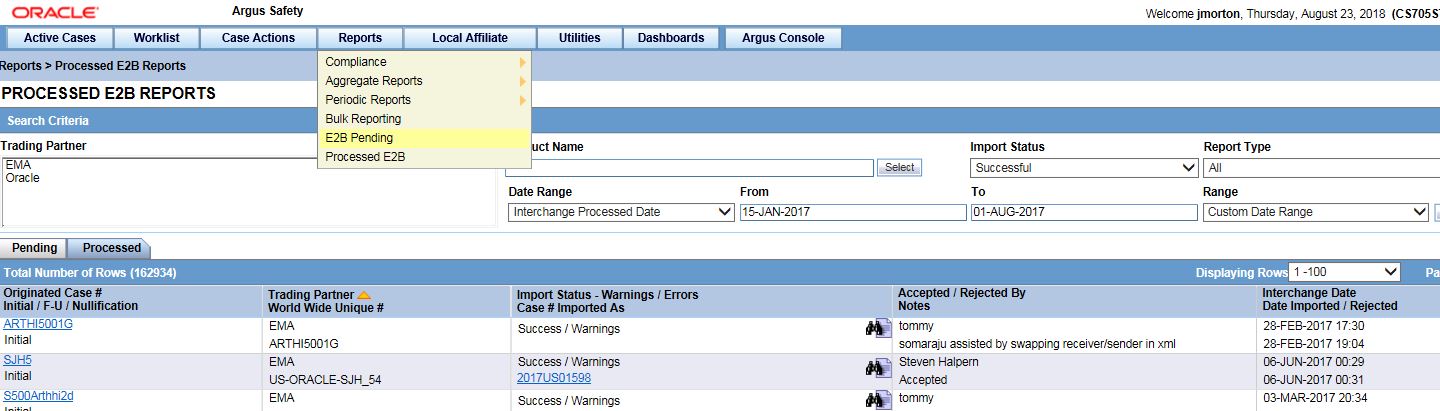
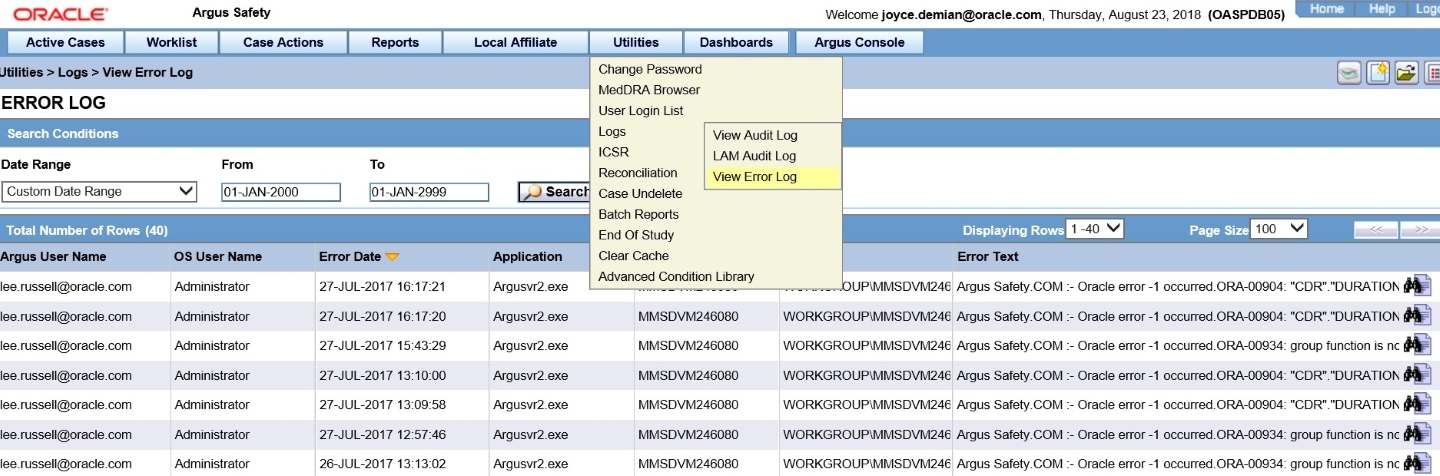
Who does this? A sponsor system administrator.
When do I do this? After the Oracle Clinical One Digital Gateway production integration has been enabled and the connection from SFTP to Argus Interchange has been made.
Follow your regular case workflow in Argus Safety. If you receive an email that a case is coming and it doesn't arrive when you expect it, contact your system administrator.
19In Argus Safety, accept or reject cases
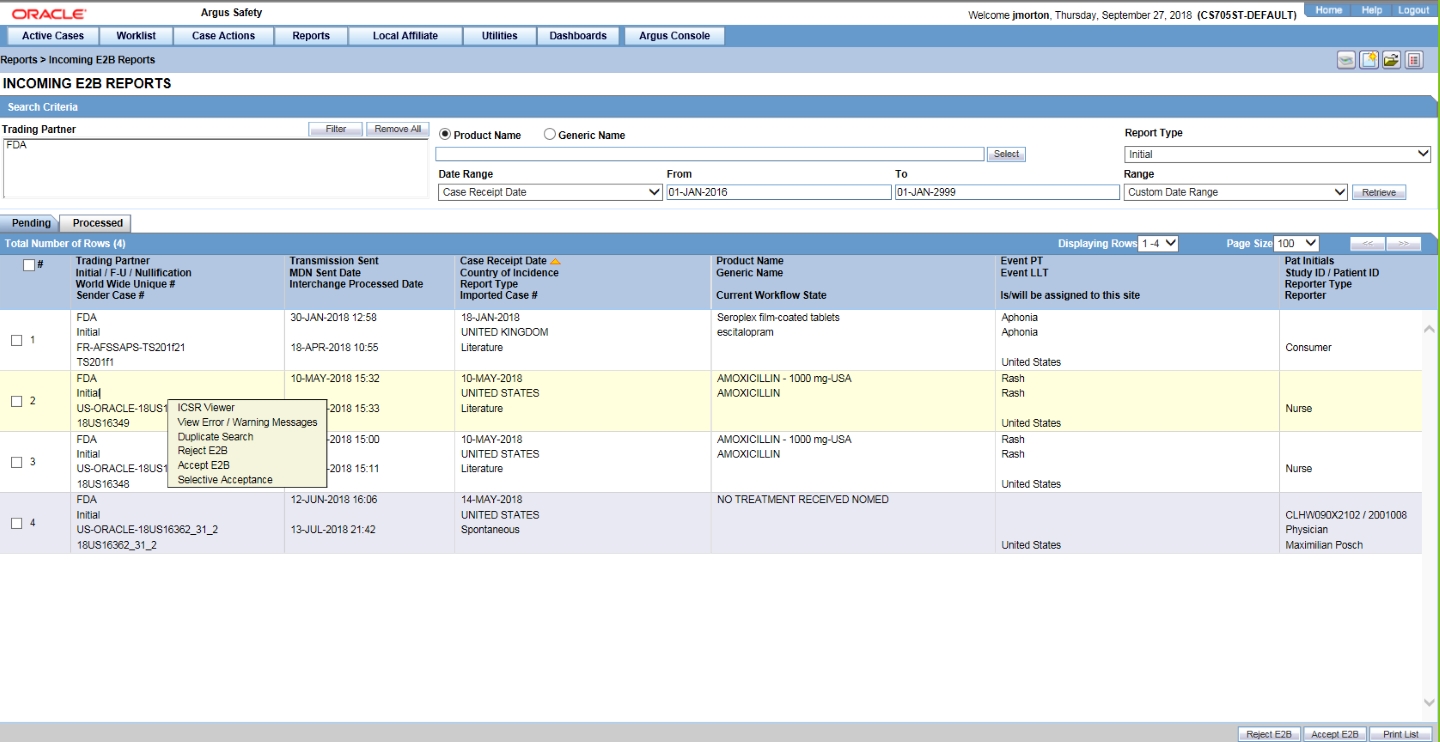
Who does this? An end user.
When do I do this? After the Oracle Clinical One Digital Gateway production integration has been enabled and the connection from SFTP to Argus Interchange has been made:
For all incoming cases, perform a duplicate search.
If you find an existing case, accept the case as a follow-up to an existing case. A new case number isn't created.
If you don't find an existing case, accept the new case. A new case number is generated, and the case is available for further processing according to your business process.
You can also reject a case. You can view all rejected cases in the incoming reports list.

 Back to Oracle Help Center
Back to Oracle Help Center You're done!
You're done!